Ja’Ceon was then given the gene therapy, a procedure that takes just minutes. 
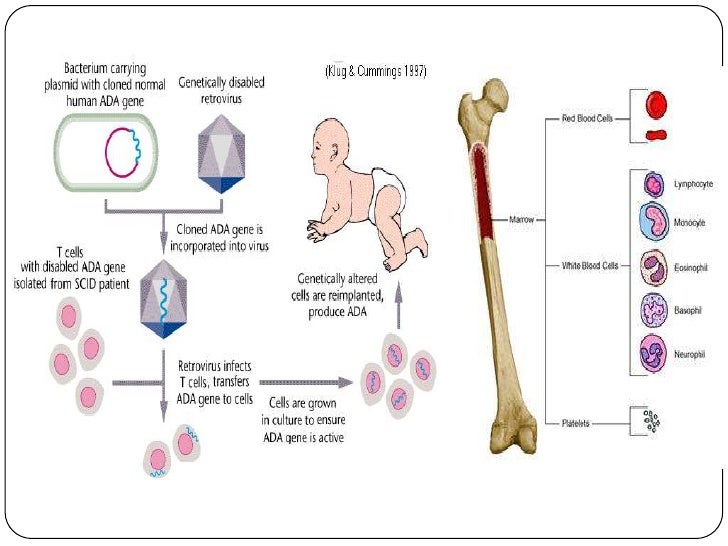
This assured there was room for the healthy cells. In the meantime, the child was given a low dose of chemotherapy. There, a virus (containing the functioning gene Ja’Ceon was missing) was combined with the stem cells from his bone marrow. He was born in New Mexico in 2016 and his SCID diagnosis was discovered with newborn screening. Ja’Ceon was one of the eight boys treated and cured with this therapy. Researchers in this new study believe the risk is minimal, but obviously follow-up for these patients is important. Despite the therapies effectiveness in treating SCID, this is clearly a dangerous side effect. In a previous study in Europe, 1/3 of participants developed the condition beginning one year after they received the therapy. The results are particularly exciting because to date, none of the children in the trial have developed leukemia. Jude will be published in the New England Journal of Medicine. All of the children have also made antibodies in response to the vaccines they’ve been given. Researchers say their immune systems are functioning just like a normal, healthy child. With fully functioning immune systems they can now live their lives like normal- attend daycare, see other children, and live life more carefree. Where are these children now? They’ve been released from the hospital where they lived their entire infancy in isolation. 
Jude for SCID in eight patients is extremely exciting. The recent success of the gene therapy treatment by St. Gene therapy is being investigated as a one-time treatment for a variety of conditions including sickle cell anemia, spinal muscular atrophy, and hemophilia as well as SCID. The healthy gene will then live and work in the body indefinitely. Gene Therapyīasically, gene therapy utilizes a virus to deliver a functioning copy of the defective gene to the patient. The company plans to file for global approval of the treatment by the end of 2021. Their therapy has successfully treated eight patients and the hospital has just announced that they have licensed the therapy to Mustang Bio. Jude Children’s Research Hospital have been working on developing a gene therapy which could serve as a potential cure for this condition. Children were kept in a sterile chamber to reduce their risk of germ exposure. That is where the term “bubble boy” comes from. At one point, those living with SCID were forced to stay in complete isolation. That means that for these patients, even a common cold can be deadly. Alpha emitter labeled anti CD-20 antibodies are promising therapeutics for NHL although a longer lived alpha emitter may be of greater efficacy.Severe Combined Immunodeficiency (SCID) is a rare disease which causes patients to practically have no immune system at all. With 55♜i 131I I-Tositumomab, 75% of the mice were tumor free by BLI and 62.5% survived.Ĭonclusion: Cure of micrometastatic NHL is achieved in the majority of animals treated at 4 days post-i.v tumor inoculation using either 213Bi rituximab or 131I tositumomab in contrast to the lack of cures with unlabeled rituximab, 90Y rituximab or if there was high tumor burden before RIT. With 100♜i 213Bi –Rituximab, 75% of the mice survived and all but one survivor was cured. With a single dose of therapy given at 4 days post iv tumor inoculation, all untreated controls and all mice in the 25♜i 90Y -Rituximab group progressed. Re-dosing of 213Bi rituximab was more effective than single dosing. Multiple in vivo studies showed significant and specific tumor growth delays with 213Bi rituximab vs free 213Bi, 213Bi control antibody and rituximab. Results: In vitro studies showed dose-dependent target-specific killing of lymphoma cells with 213Bi Rituximab. 213Bi Rituximab was also compared to the low energy beta-emitter 131I I-Tositumomab and the high energy beta emitting 90Y - Rituximab in vivo. Single and multiple dose regimens were explored and results with 213Bi Rituximab compared to various controls including no treatment, free 213Bi radiometal, unlabeled Rituximab, and 213Bi-labeled anti-HER2/neu (non-CD20 specific antibody). In vitro and in vivo radioimmunotherapy experiments were conducted. 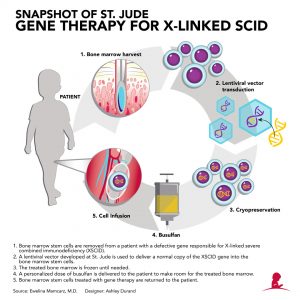
Methods: A highly-lethal SCID mouse model of minimal tumor burden disseminated NHL was established using human Raji lymphoma cells transfected to express the luciferase reporter. Objectives: We studied the feasibility of alpha emitter, 213Bi -Anti-CD20 therapy with direct bioluminescent tracking of micrometastatic human B-cell Lymphoma in a scid Mouse Model.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
